Ultrasound Imaging and Ultrasound-Guided Injection of Facial Aesthetic Injectables: A Comprehensive Review
Introduction
Minimally invasive cosmetic procedures have increased in popularity over the last decade, driven by safer and more effective dermal fillers for facial rejuvenation.1 Injectable fillers can restore volume, soften wrinkles, and improve skin texture with minimal downtime. These effects are achieved through two main mechanisms: (1) physical volumization, via space-filling and water-binding properties, as seen in hyaluronic acid (HA) gel; and (2) biostimulation of new collagen production using agents such as poly-L-lactic acid (PLLA) or calcium hydroxylapatite (CaHA).2
Ultrasound imaging has recently emerged as a game-changing tool in filler treatment. High-frequency (e.g., 15–20 MHz) ultrasound can visualize soft tissues at high resolution and identify fillers in situ, distinguish tissue planes, and even detect blood vessels with Doppler mode.3 Real-time ultrasonography provides visual guidance during injection, enhancing the accuracy of filler placement and helping to avoid vital anatomical structures. This is especially valuable in high-risk facial regions (such as the temple, forehead/glabella, and nose), where inadvertent intravascular injections can cause severe complications (skin necrosis, blindness, and stroke). Moreover, ultrasound can aid in the diagnosis and management of filler-related adverse events, including differentiating granulomas from abscesses and precisely locating filler deposits to guide hyaluronidase injection. This review provides a comprehensive overview of facial aesthetic fillers with an emphasis on ultrasound imaging findings and ultrasound-guided techniques.
Hyaluronic Acid Fillers (e.g., Juvederm): HA-based fillers are the most widely used fillers due to their versatility, immediate results, and reversibility with hyaluronidase.
Hybrid Cooperative Hyaluronan (Profhilo): Profhilo® is a next-generation HA injectable intended for skin bio-remodeling rather than volumizing or contouring. Clinical studies have demonstrated significant improvements in skin hydration, elasticity, and overall skin quality after Profhilo treatments, with high patient satisfaction.4 Ultrasonographically, Profhilo is anticipated to behave similarly to other non-cross-linked HA gels: initially anechoic on injection, followed by gradual tissue integration without the persistence of visible nodules. Nonetheless, published imaging studies on Profhilo are currently limited.
Collagen-Stimulating Fillers (PLLA/Sculptra and CaHA/Radiesse): Biostimulatory fillers induce a controlled foreign body response that results in neocollagenesis over time.
Calcium hydroxylapatite (CaHA) filler: Radiesse® consists of synthetic CaHA microspheres (25–45??m in diameter), comprising 30% of the product by volume, suspended in a 70% CMC/water gel carrier.5 A split-arm trial6 (injecting PLLA in one forearm vs. diluted CaHA in the other) found that both resulted in comparable collagen deposition; however, CaHA-treated tissue showed 6-fold greater elastin fiber formation and fewer foreign body giant cells than did PLLA, potentially contributing to superior clinical outcomes in skin elasticity.
KARISMA (Collagen/HA Biostimulator): KARISMA® represents a recent advancement in collagen-based dermal fillers, formulated with recombinant human type I collagen polypeptide, high molecular weight HA, and carboxymethylcellulose (CMC). To date, ultrasound appearances specific to KARISMA have not been previously published. However, it can be hypothesized to display imaging characteristics similar to those of other collagen-based fillers—namely, a homogeneously hypoechoic implant that becomes isoechoic as exogenous collagen integrates into the surrounding tissue. The presence of CMC may initially generate reflective echoes, as CMC (a cellulose derivative) has been associated with hyperechoic signals and localized fibrosis (Figure 1).
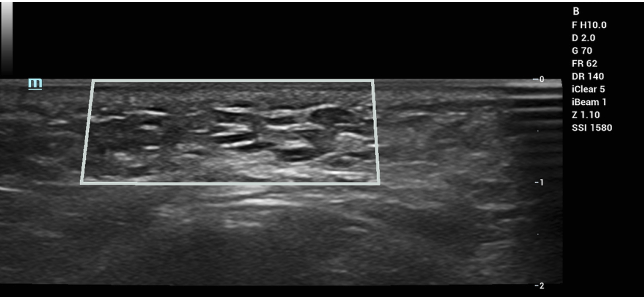
Figure 1: Ultrasound imaging of KARISMA (3 days after injection). Imaging revealed multiple hypoechoic implants with a peripheral hyperechoic ring of carboxymethylcellulose.
Ultrasound Characteristics of Fillers in Vivo
High-frequency ultrasound provides a non-invasive window for assessing filler location, distribution, and tissue response. Different filler materials exhibit distinctive echogenic patterns on ultrasound. In a seminal ultrasound study by Urdiales-Gálvez et al., four basic patterns were described1 (Figure 2):
- Heterogeneous pattern: a mix of hyperechoic and anechoic areas interspersed in the tissue. This appearance is typical of normal skin, subcutaneous fat, and, in fully integrated HA fillers, sometime post-injection. Essentially, once an HA gel has partly resorbed and infiltrated the surrounding tissue, it no longer appears as distinct fluid pockets but rather as a subtle, mottled increase in echogenicity within the soft tissue. A successfully integrated HA filler may be almost indiscernible from the normal subcutis on ultrasound.
- Globular (cystic) pattern: well-defined anechoic or hypo-echoic “cyst-like” areas often with posterior acoustic enhancement (bright echo deep to the void). This indicates a liquid or semi-liquid material and is characteristic of fresh HA deposits immediately after injection. Free injectable silicone (a permanent filler) also shows anechoic globules but usually with some scattering. Appear as localized, dark, non-echogenic areas reflecting the presence of HA gel boluses, sometimes bordered by a thin capsule. Polyacrylamide hydrogels, due to their water-rich composition, may present with a comparable anechoic profile.
- Coarse-grain “snowfall” pattern: numerous small discrete hyperechoic foci scattered throughout the tissue, often without a heavy posterior shadow (as the material is less infiltrative than silicone). This pattern is typical of particulate fillers, such as CaHA and polymethylmethacrylate (PMMA). The hyperechoic “snowflakes” are evenly distributed if the filler was well placed. Over time, as collagen forms around these particles, the overall echogenicity can further increase (because collagen is echogenic). The fine-grain snowfall pattern consists of minute hyperechoic points accompanied by comet-tail artifacts and strong acoustic shadowing. It is typically associated with liquid silicone injections, where the numerous microdroplets generate reverberation artifacts. Silicone patterns can obscure deeper structures, making the teeth difficult to visualize in lip silicone cases. CaHA’s echoes are coarser and typically do not produce as much reverberation as those of silicone.
- Heterogeneous fibrous pattern with hyperechoic strands: This pattern has been observed in association with fillers that strongly induce fibrosis (such as CaHA, PLLA, or polycaprolactone). These biostimulatory fillers often result in increased overall echogenicity of the treated area over time. In other words, months after the injection of a biostimulator, a uniformly brighter (whiter) area may appear in the dermis or subcutis compared to the surrounding untreated tissue, corresponding to newly formed collagen. Urdiales-Gálvez et al. reported that collagen-inducing materials, such as CaHA and PLLA, showed more pronounced hyperechoic bands in the tissue than did purely hydrating fillers, such as HA.
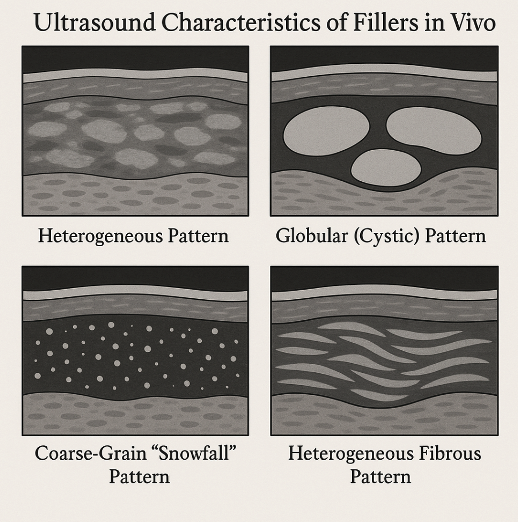
Figure 2: Ultrasound Characteristics of Fillers in Vivo.
Utrasound imaging of facial fillers provides real-time feedback on filler placement (confirming whether the product is in the intended plane), filler type characteristics (gel vs. particulate), and tissue reaction (fibrosis and vascularity). It can help identify complications such as unintentional intramuscular or intravascular injections, asymmetric filler distribution, or residual material from prior injections. These insights underscore why ultrasound is increasingly being adopted by advanced injectors and is an area of active research in aesthetic medicine.
Ultrasound-Guided Injection Techniques
Traditional filler injections rely on practitioners’ anatomical knowledge and tactile feedback. Ultrasound guidance introduces direct visualization into this process, potentially redefining injection safety standards. Two principal approaches have been described: pre-injection scanning and real-time guidance.
• “Scan before injecting”: The clinician uses ultrasound to map the anatomy of the treatment area immediately prior to injection. Critical structures, such as arteries or veins, can be identified and marked on the skin (e.g., marking the course of the temporal artery in the temple region). Ultrasound mapping is especially valuable in patients with prior procedures where filler or scar tissue from past treatments may be present.
• “Scan while injecting”: In this technique, the injection is done under direct ultrasound visualization, typically using an in-plane approach so the entire cannula/needle and filler flow can be seen in real time. Additionally, it allows for real-time assessment of tissue expansion, aiding in optimal filler placement.
Another novel technique is ultrasound-guided aspiration and dissolution: if arterial occlusion is suspected, the provider can immediately scan, locate the intravascular filler, and attempt to aspirate or inject hyaluronidase under guidance. This approach has been used to treat impending necrosis in the nose and glabellar region, potentially improving outcomes by delivering antidotes precisely into the occluded vessel or area of ischemia. Ultrasound guidance is also useful in chronic complications such as nodules, enabling accurate needle insertion for intralesional steroid delivery, enzymatic degradation with hyaluronidase (in the case of HA-based fillers), or mechanical disruption.
Learning curve and practicality: Mastering ultrasound-guided injections requires training, and the injector must be adept at interpreting ultrasound images and coordinating the probe and needle simultaneously. Initially, these procedures take more time to perform. A common strategy is to perform thorough ultrasound mapping (pre-scan), followed by injection without continuous scanning, using the anatomical knowledge acquired during the pre-scan. As competency grows, real-time injection can be reserved for the highest-risk areas (e.g., fillers to the nose, where blindness is a concern, since the dorsal artery should be avoided and the filler should be placed precisely above the periosteum).
Safety Considerations
The aspiration technique is a traditional safety step, but its reliability is debated; small needles can be occluded by tissue, resulting in false negatives. Ultrasound is more accurate than needle aspiration in determining whether a needle tip is inside a vessel or not.7 Proper patient counseling about signs of occlusion (pain, color change) and maintaining a readily accessible emergency kit (hyaluronidase, aspirin, etc.) are essential safety measures. Layering techniques (placing small aliquots in multiple tissue planes) often increase safety and natural appearance. Ultrasound facilitates this approach by providing clear visualization of each anatomical layer. Moreover, it supports accurate documentation of injection sites, which is beneficial for legal records and future treatment planning—functioning much like a three-dimensional map of previous filler placements, particularly useful when care is transferred between clinicians.
Future outlook: As ultrasound usage becomes more widespread, it may lead to standardized protocols for guided injections. This could conceivably reduce complication rates across the board and allow for the more adventurous yet safe use of long-lasting biostimulators in previously avoided areas. The use of dilute CaHA for tear trough rejuvenation, guided by ultrasound, presents a potential alternative to HA fillers by offering enhanced longevity and biostimulatory effects. While currently off-label and limited by the risk of nodules, this technique may be implemented safely by experienced injectors with the aid of real-time imaging.
Another development is combination treatments evaluated with ultrasound, for example, combining fillers with autologous treatments (PRP or fat grafting) to observe synergistic effects. In a review by Humzah et al.,4 an interesting concept was the addition of Profhilo (HA) to fat grafts to improve fat survival, with ultrasonography and histology confirming better fat retention and vascularity. Such approaches herald a new era of regenerative aesthetics, where fillers are not just “fillers” but agents to improve tissue health.
Conclusion
Facial aesthetic injectables have evolved from purely volumizing agents to tools that can improve skin physiology and induce tissue regeneration. When used judiciously, with the aid of ultrasound imaging and guided techniques, these fillers are efficacious in reversing signs of aging with a high safety profile. Ongoing research, including prospective comparative trials and long-term ultrasound monitoring studies, is anticipated to provide greater insight into optimal practices. Collaboration among dermatologists, plastic surgeons, and radiologists in this arena is particularly exciting, as it fosters a multidisciplinary understanding of how to best use imaging to augment clinical artistry. Practitioners who embrace evidence-based innovations are likely to deliver superior, consistent, and safer patient outcomes. The integration of ultrasound into aesthetic medicine exemplifies the field’s progress toward more precision medicine, ensuring that the pursuit of beauty does not come at the expense of safety.
Abbreviations
HA, Hyaluronic Acid; PLLA, Poly-L-lactic Acid; CaHA, Calcium Hydroxylapatite; BDDE, 1,4-Butanediol Diglycidyl Ether; G?, Elastic Modulus; CMC, Carboxymethylcellulose; TGF-?, Transforming Growth Factor Beta?References
1. Urdiales-Gálvez F, De Cabo-Francés FM, Bové I. Ultrasound patterns of different dermal filler materials used in aesthetics. J Cosmet Dermatol. 2021;20(5):1541-1548. doi: 10.1111/jocd.14032.
2. Guo J, Fang W, Wang F. Injectable fillers: Current status, physicochemical properties, function mechanism, and perspectives. RSC Adv. 2023;13(34):23841-23858. doi: 10.1039/d3ra04321e.
3. Sigrist R, Desyatnikova S, Chammas MC, Vasconcelos-Berg R. Best practices for the use of high-frequency ultrasound to guide aesthetic filler injections-Part 1: Upper third of the face. Diagnostics (Basel). 2024;14(16):1718. doi: 10.3390/diagnostics14161718.
4. Humzah D, Molina B, Salti G, Cigni C, Bellia G, Grimolizzi F. Intradermal injection of hybrid complexes of high- and low-molecular-weight hyaluronan: Where do we stand and where are we headed in regenerative medicine? Int J Mol Sci. 2024;25(6):3216. doi: 10.3390/ijms25063216.
5. Aguilera SB, McCarthy A, Khalifian S, Lorenc ZP, Goldie K, Chernoff WG. The role of calcium hydroxylapatite (Radiesse) as a regenerative aesthetic treatment: A narrative review. Aesthetic Surg J. 2023;43(10):1063-1090. doi: 10.1093/asj/sjad173.
6. Mazzuco R, Evangelista C, Gobbato DO, de Almeida LM. Clinical and histological comparative outcomes after injections of poly-l-lactic acid and calcium hydroxyapatite in arms: A split side study. J Cosmet Dermatol. 2022;21(12):6727-6733. doi: 10.1111/jocd.15356.
7. Schelke LW, Decates TS, Velthuis PJ. Ultrasound to improve the safety of hyaluronic acid filler treatments. J Cosmet Dermatol. 2018;17(6):1019-1024. doi: 10.1111/jocd.12726
Etiquetas: Inyecciones
Share this article on


