FEATURES OF PATHOGENETIC MECHANISMS OF SKIN HYPERPIGMENTATION: A ONE-MOMENT CROSS-SECTIONAL RESEARCH
Introduction
There are no uniform statistics on primary acquired hyperpigmentation (melasma), various authors estimate its prevalence from 1% in the general population to 50% in high-risk groups. According to medical statistics, women aged 25 to 50 with skin phototypes 3-6 are most susceptible to developing melasma [1,2]. The average ratio of men and women with such changes is 1:9 [2]. Skin color is determined by the presence of pigments - melanin and carotenoids, and also depends on the level of hemoglobin in the blood. Despite the uniqueness of the melanin pigment system, when the final outcome of pigmentation is formed as a result of the interaction of two types of cells (melanocytes and keratinocytes), researchers have currently achieved some success in partially understanding the mechanism of melanogenesis and treating patients. Undoubtedly, pigmentation disorders associated mainly with melanin are of greatest importance for clinical practice.
Melanin synthesis occurs in highly specialized epidermal cells called melanocytes, which are of neurogenic origin and morphologically similar to nerve cells. Melanocytes are located among the cells of the basal layer and have several processes that can extend up to 100 µm from the cell body. The end of the melanocyte branch after repeated division is closely adjacent to the keratinocyte pole. The distribution of melanocytes and the way they branch are normally such that there is hardly a cell of the basal layer that does not contact the terminal sections of the dendrites. There is also contact between the processes of melanocytes. The structural and functional association of the melanocyte with keratinocytes is called the "epidermal-melanin unit of the epidermis". Melanin synthesis occurs in melanocytes in special organelles called melanosomes, which are quite complex formations. They are surrounded by a continuous membrane and contain a highly organized internal structure of longitudinally oriented strands or concentric plates. Melanosomes can be spherical or ellipsoid and have a size of 0.5-1 micrometre. In their development, melanosomes go through 4 stages and, moving towards the periphery of the pigment cell, acquire an ever-increasing electron-optical density until their structure ceases to differ. In this form, they are transferred to keratinocytes (a cytocrine method similar to the production of secretion in other organs, but now the transfer of melanin is considered a phagocytic process). The biochemical mechanisms of melanin synthesis are well studied. From the amino acid tyrosine, under the influence of tyrosinase of melanocytes, dihydroxyphenylalanine is formed and through a number of intermediate compounds is converted into melanin, which in the body exists in the form of a compound with protein - melanoprotein complex [1, 2].
During the development of an inflammatory reaction in the epidermis, alpha-arachidonic acid is oxidized to prostaglandins and leukotrienes, and reactive oxygen species are generated. Presumably, eicosanoids (leukotrienes, prostaglandins), cytokines and inflammatory mediators (interleukins, tumor necrosis factor-alpha, etc.) can affect melanocytes, increasing the level of immunoreactive tyrosinase, which stimulates hyperplasia and/or hypertrophy of melanocytes, melanogenesis and/or activation of melanin transfer to surrounding keratinocytes [3].
When the basement membrane and basal keratinocytes are damaged, the melanin they contain enters the upper layers of the dermis. There, it is absorbed by macrophages, which migrate to the foci of inflammation [4].
The concept of melanogenesis regulation is sufficient to understand the multi-stage nature of this process. Some literary sources indicate a direct influence of catecholamines on melanogenesis through the hypothalamus, which stimulates the pituitary gland to produce melanocyte-stimulating hormone [5]. An important role in the mechanism of melanogenesis belongs to the activity of endocrine glands: the pituitary gland, pineal gland, adrenal cortex, thyroid gland and sex glands. The mechanism of melanogenesis is due to a complex biological process involving tyrosine, tyrosinases, copper ions, zinc, and molecular oxygen [1,2].
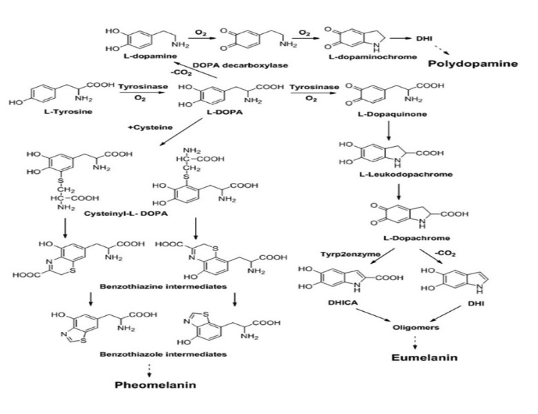
Fig.1. Melanogenesis.
Microelements, especially copper, play an important role in the mechanism of melanogenesis disorders [2,6]. It has been established that with a significant decrease in copper content in depigmented skin, its content in the blood and daily urine increases, which is apparently associated with a violation of copper absorption by tissues. Zinc is involved in one of the stages of melanin synthesis in the process of tyrosine oxidation. The participation of the immune system in the mechanism of melanogenesis is being studied. Morphological studies by various authors have established a change in the number of melanocytes and their dystrophy in foci of dyschromia in the direction of increase or decrease. In recent years, assumptions have been made about the participation of radical chain reactions in the processes of pigment formation. In this regard, the state of protection against oxygen radicals, including enzymes, is important: superoxide dismutase, catalase, glutathione, peroxidase, as well as natural antioxidants - tocopherols, ascorbic acid, ubiquinones. They are part of a system that prevents uncontrolled radical reactions. Analysis of skin pigmentation under the influence of PUVA therapy confirms the idea that melanin is formed during the tyrosinase reaction due to oxidation or blocking of sulfhydryl groups. Tyrosinase is activated by UVR by destroying peptidase and glutathione, which block tyrosinase.
Some studies have also noted pronounced solar elastosis-like changes in melasma lesions and suggested a cumulative effect of ultraviolet radiation: induction of inflammatory cytokines such as interleukin-1, endothelin-1, alpha-melanocyte-stimulating hormone (alpha-MSH), adrenocorticotropic hormone (ACTH) leads to stimulation of melanocytes and the appearance of pigmentation [2,4].
Another supposed mechanism for the development of melasma is an increase in the level of vascular endothelial growth factor (VEGF) and increased vascularization (an increase in the size and number of vessels) in the lesions. Considering that human melanocytes normally express VEGF receptors on their surface, this indicates the ability of melanocytes to respond to angiogenic factors, and therefore explains the role of the vascular component in the development of melasma. It should be noted that vascular endothelial cells can produce cytokines, plasminogen and other factors that also affect melanogenesis [7].
Some publications note the role of hormonal influence, which is based on increased expression of receptors to estrogen and progesterone in pigmented areas. These hormones have the ability to stimulate melanogenesis through DOPAchrome tautomerase and tyrosinase. Activation of tyrosinase can be carried out by hormones directly or indirectly (through an increase in the level of cAMP).
It is known that one of the clinical signs of skin hyperpigmentation is localized hyperkeratosis in the lesion, associated with a high level of cell proliferation and saturation of cells with melanin. Cell proliferation and hyperkeratosis are associated with an increased need for lipids in the epidermis. It is known that the transport form of lipids in the blood are lipoproteins of various classes. They transport lipids to tissues for the construction of cell membranes, the synthesis of steroid hormones and fatty acids to maintain cellular metabolism. However, it is important to emphasize that the composition of lipoproteins circulating in the blood is not static. They are in a dynamic state with a constant exchange of components between different types. Plasma lipoproteins transport metabolites, hormones, vitamins, maintain cholesterol homeostasis, so their functional and physicochemical state is an important link in assessing the adaptive capabilities of the body. Changes in the ratio of different lipoprotein fractions can become a predictor of the formation of skin hyperpigmentation. Literature data indicate that certain classes of lipoproteins, such as HDL, can help maintain vascular endothelial function by stimulating the release of nitric oxide (NO) [8]. The ability of melanocytes to respond to angiogenic factors is known. Vascular endothelial cells can produce cytokines, plasminogen, and other factors that also affect melanogenesis.
The aim of the research: to study the level of lipoproteins of various classes in patients with hyperkeratosis and their role in the formation of the pathogenesis of skin hyperpigmentation.
Materials and methods. The research included 50 women living in Rostov-on-Don, average age 42.3±1.1 years, divided into two groups: 25 people with skin hyperpigmentation, average age 41.52±1.68 years, undergoing treatment in the dermatology department; 25 people - practically healthy (PH) individuals - during medical examinations, average age 43.57±1.43 years. The examination of all participants was carried out after signing informed consent.
The content of total cholesterol, TG, HDL was determined by the enzymatic colorimetric method using the ChronolabAG kits (Switzerland), LDL - by the turbidimetric method, very low density lipoproteins (VLDL) - by the TG/5 calculation method. Statistical processing of the obtained data was carried out using Microsoft Office Excel 2007 (Microsoft Corp., USA) and Statistica 10.0 (StatSoft Inc, USA). The data are presented as M ± m, where M is the average value of the feature, m is the average error of the feature, as well as an expression of the frequency of occurrence of the feature in absolute values (n) and percentages (%). The significance of differences between the subgroups was assessed using Student's T-test. Correlation analysis was carried out to determine the relationships between different parameters. The threshold level of statistical significance was p < 0.05. The Mann-Whitney test was used for comparative analysis of absolute differences between samples.
Results and discussion
During the study of the metabolic features of lipoprotein metabolism in patients with hyperpigmentation, an increase in the level of individual lipoprotein classes was noted compared to the control group. The level of total cholesterol, triglycerides, LDL, VLDL was statistically significantly increased, and a tendency towards a decrease in HDL was revealed. Thus, the content of TC in patients with hyperpigmentation was statistically significantly different from the control, by 18.64% (p = 0.148), which suggests that cholesterol is used to build cell membranes. Against this background, a higher content of TG was noted - by 18.1% (p = 0.393), which are obviously used to maintain cell energy costs. It should be noted that the concentrations of atherogenic fractions of VLDL, LDL in hyperpigmentation were higher than in the control: VLDL - by 18.13% (p = 0.352) and LDL - by 44.93% (p = 0.044). Which is obviously associated with increased cholesterol esters entering the epidermal tissue. However, HDL values tended to decrease in the main group compared to the control. HDL have a number of biological properties, including antioxidant and cytoprotective.
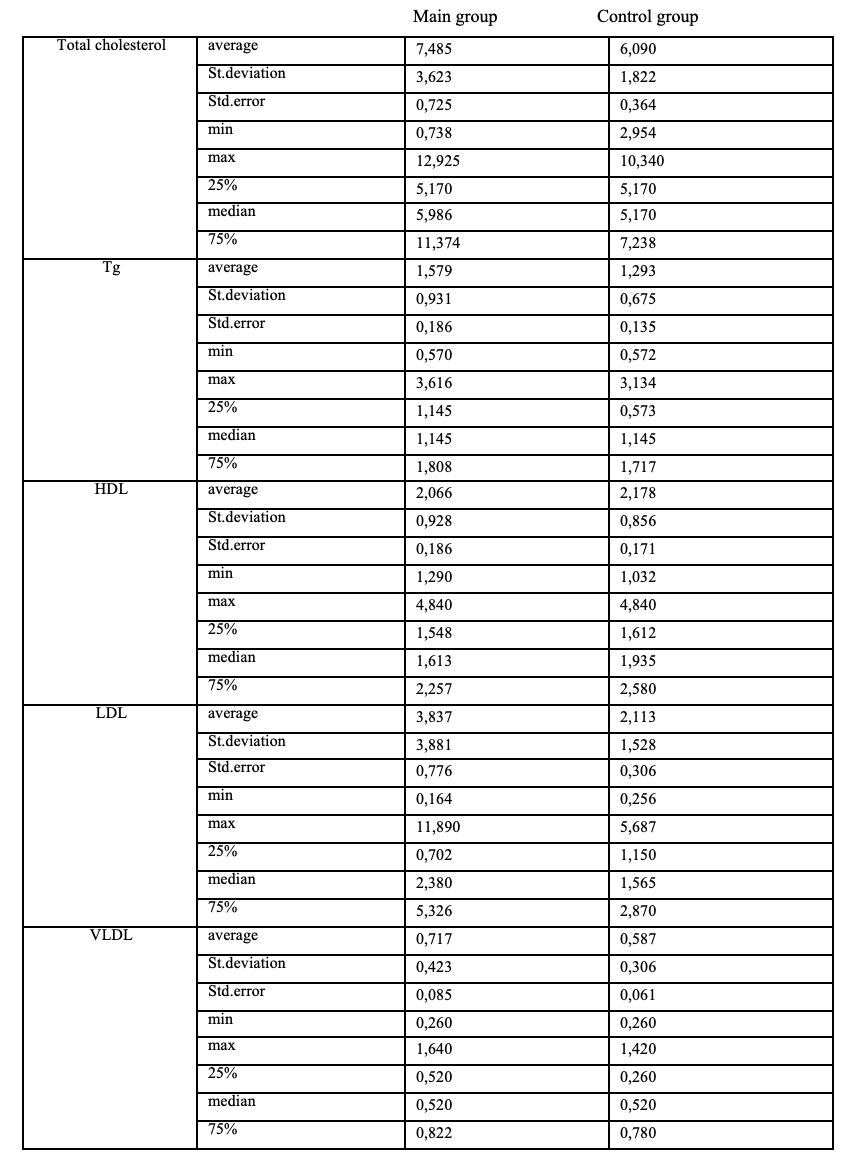
Table 1. Contents of “traditional” lipid metabolism parameters in patients (descriptive statistics)
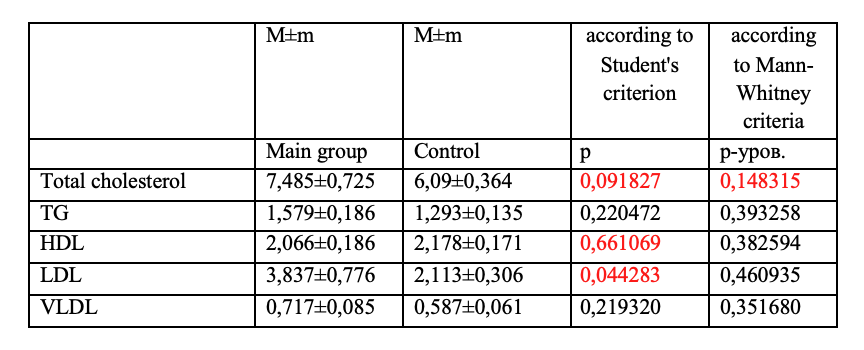
Table 2. Comparative analysis. Statistically significant differences are highlighted in red
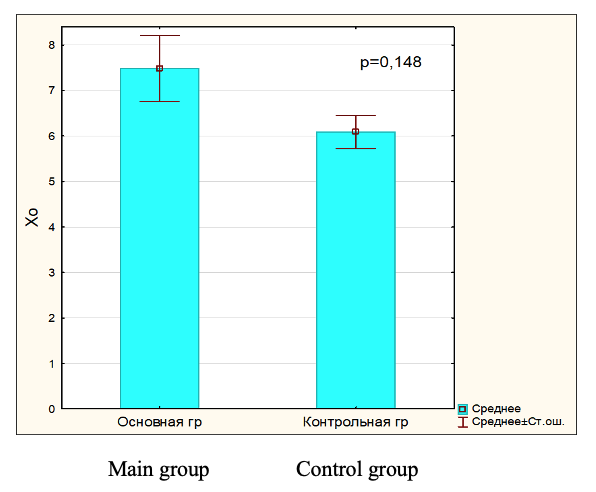
Fig.2. Total cholesterol in blood serum in main and control group
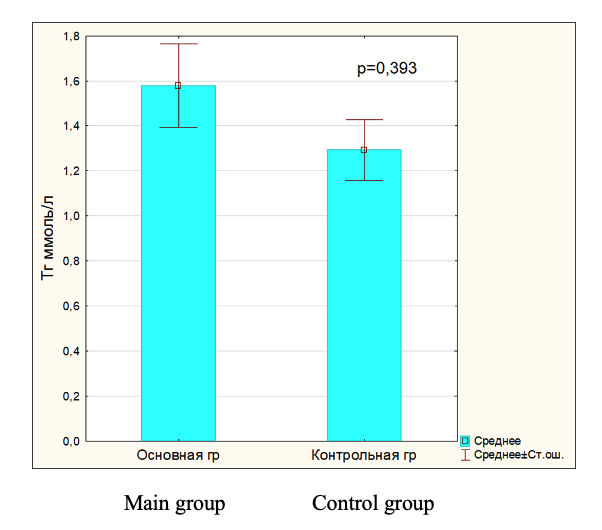
Fig.3. Triglyceride level in blood serum in main and control group.
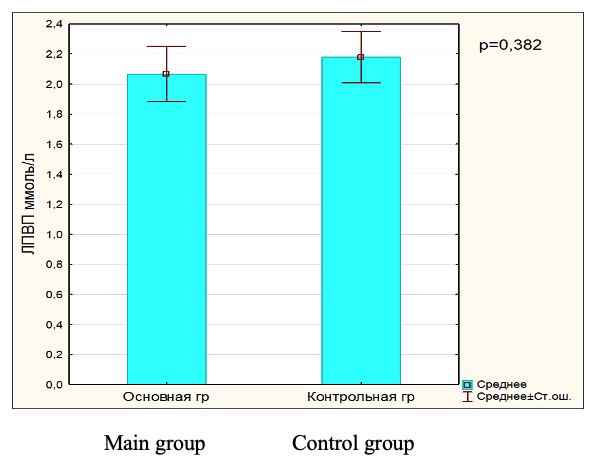
Fig.4. HDL level in blood serum in main and control group.
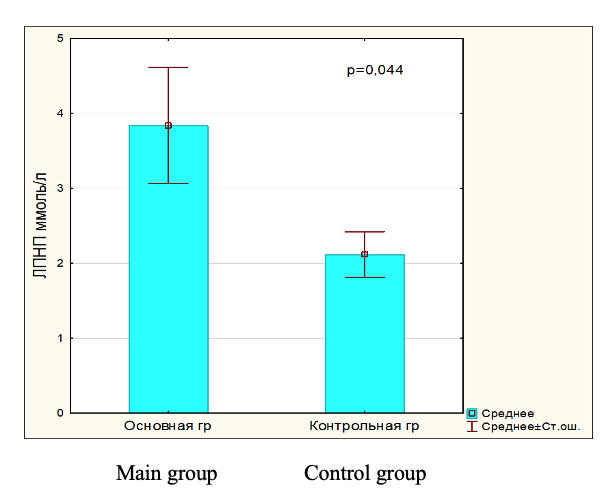
Fig.5. LDL level in blood serum in main and control group.
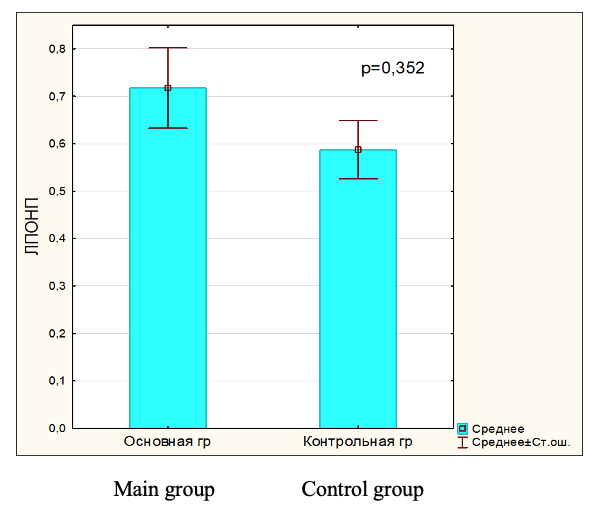
Fig.6. VLDL level in blood serum in main and control group.
Conclusion
Thus, analyzing the obtained results, we found that patients with hyperpigmentation have statistically significantly increased levels of individual classes of lipoproteins, such as LDL, VLDL, as well as triacylglycerides and total cholesterol. This is probably an indicator of the constant need of cells to synthesize membrane lipids and maintain increased cellular metabolism, which is necessary for increased regeneration of the epidermis. Further study of the level of blood plasma lipoproteins and analysis of changes in their fractions will allow us to study the pathogenetic mechanisms of skin hyperpigmentation, as well as to reveal the predictors that contribute to its formation. The feasibility of studying the systemic biochemical profile in these patients will allow us to develop optimal drug correction regimens.
REFERENCES
1. Kubanova A.A., Manturova N.E, Gallyamova Y.A. Manual on cosmetology. ANO Publishing House "Scientific Review", 2020 - 728.
2. Potekaev N.N., Avram M.R., Tsao S., Tannous Z., Avram M.M. Atlas of Cosmetic Dermatology / translated from English. Publishing House Binnom, 2013: 295.
3. Burylina O.M., Karpova A.V. Cosmetology: Clinical Guide. Geotar-Media 2018: 744.
4. Torres-Alvarez, B., Mesa-Garza, I.G., Castanedo-Cazares, J.P., Fuentes-Ahumada, C., Oros-Ovalle, C., Navarrete-Solis, J., and Moncada, B. (2011). Histochemical and immunohistochemical study in melasma: evidence of damage in the basal membrane. Am. J. Dermatopathol. 33, 291-295.
5. Kim, N.H., Lee, C.H., and Lee, A.Y. (2010). H19 RNA downregulation stimulated melanogenesis in melasma. Pigment Cell Melanoma Res. 23, 84-92.
6. Cardinali, G., Kovacs, D., Giglio, M.D., Cota, C., Aspite, N., Amantea, A., Girolomoni, G., and Picardo, M. (2009). A kindred with familial progressive hyperpigmentation-like disorder: implication of fibroblast-derived growth factors in pigmentation. Eur. J. Dermatol. 19, 469-473.
7. Kim, E.H., Kim, Y.C., Lee, E.S., and Kang, H.Y. (2007). The vascular characteristics of melasma. J. Dermatol. Sci. 46, 111-116.
8. Klimov A.N., Parfenova N.S., Golikov Yu.P. To the 100th anniversary of the creation of the cholesterol model of atherosclerosis. Biomedical Chemistry.2012;58:1:5-11.
Etiquetas: Dermatología clínica & cirugía dermatológica
Share this article on


