Biological Effects of HA Based Dermal Fillers and Fractional Ablative Laser Therapy Full-Thickness Skin Equivalents
Background
Injection of dermal fillers is one of the most frequently performed aesthetic procedures. Hyaluronic acid (HA) fillers are commonly used due to their hygroscopic property, biocompatibility and reversibility. So far, the direct biologicals effects of dermal fillers monotherapy and combination therapy with ablative fractional CO2 - or Er:YAG laser irradiation on human skin cells, especially epidermal keratinocytes and dermal fibroblasts are not completely understood. To avoid the need for sequential invasive skin biopsies from patients or the implementation of animal testing, human organotypic three-dimensional (3D) skin equivalents have been established as an appropriate alternative for standardized studies of the human skin. This study aimed at understanding more precisely the biological effects of HA-based fillers, their combination with laser treatment and their differences to poly-l-lactic acid (PLLA)-based fillers.
Material and Methods
Experiments were performed on human 3D full-thickness skin equivalents. Different stabilized HA and PLLA-based fillers were injected intradermally into the dermal equivalent of a skin model as a monotherapy and in combination with ablative fractional CO2 - or Er:YAG laser irradiation. Biological effects on skin morphology (histological analysis) and gene expression (microarray analysis) were investigated at different time points (d0, d5) after treatment.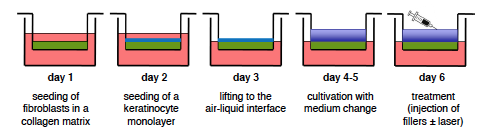
Results
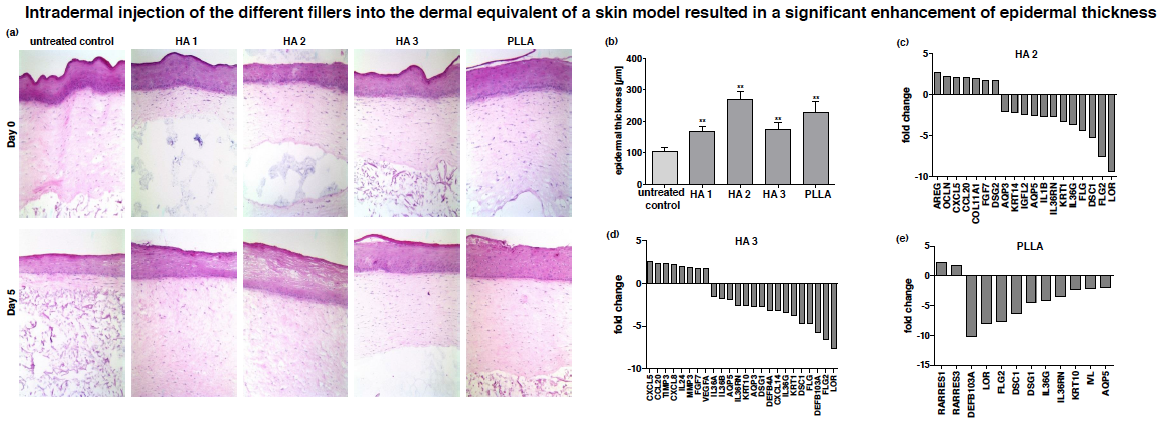
Figure 1. (a) Representative HE stained sections of 3D skin models on day 0 and day 5 after intradermal injection of different HA-based fillers (HA 1: 20mg/ml Non-Animal Stabilized Hyaluronic Acid (NASHA) gel without particles; HA 2: 12mg/ml NASHA gel with fine particles; Ha 3: 20mg/ml NASHA gel with bigger particle size) and a poly-l-lactic acid (PLLA)-based filler. (b) Measurement of epidermal equivalent thickness on day 5 after injection. (c-e) Gene expression profiling in models that were injected with HA 2, HA 3 and PLLA on day 5 after injection (Affymetrix ®
Gene Chip Human Exon 2.0 ST array).
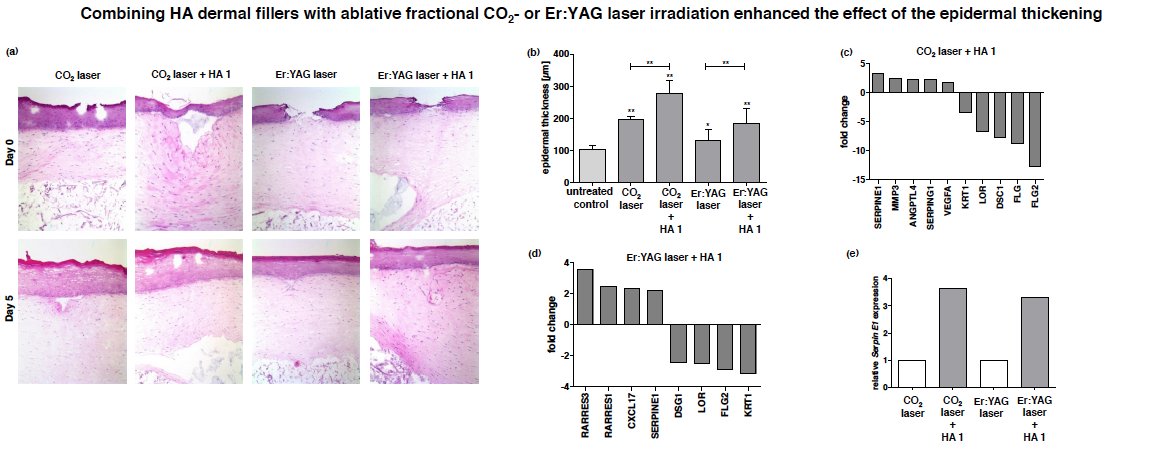
Figure 2. (a) Representative HE stained sections of 3D skin models on day 0 and day 5 after ablative fractional laser treatment with either a CO2 or an Er:YAG laser and a combination of both laser treatments with intradermal injection of HA filler 1 (20mg/ml NASHA gel without particles ). (b) Measurement of epidermal equivalent thickness on day 5 after treatment. (c-d) Gene expression profiling in models that were treated with a combination of laser irradiation and intradermal injection of HA 1 on day 5 after treatment in contrast to laser irradiated models (Affymetrix ® Gene Chip Human Exon 2.0 ST array). (e) Independent Real-Time PCR analysis of Serpin E1 on day 5 after treatment.
Conclusions
• Our data show enhancing effects of dermal fillers on epidermal thickness and prove the proliferating effects of these products on epidermal cells on the molecular level.
• Gene expression profiling revealed an upregulation of modulators of tissue remodeling (e.g. TIMP3, SERPIN E1) and collagens (COL11A1) as well as a downregulation of differentiation markers (e.g. FLG, LOR, KRT1) and proinflammatory cytokines (e.g. IL-36, IL-1).
• HA-derived products revealed a specific upregulation pattern of chemokines such as CXCL5 and CCL20, in contrast to poly-l-lactic acid-based injectable dermal fillers. These data suggest a secondary effect of these HA-based compounds on the immune cells of the skin, especially monocytes and macrophages.
• Our findings clearly emphasize supporting effects of fractional ablative laser treatment on HA dermal fillers injection and suggest a combination of both treatments.
Mots-clés: Dermatologie clinique & chirurgie dermatologique, Injectables, Lasers, EBD et remodelage du corps
Partagez cet article sur



