This sponsored article is a contribution from QATAR PHARMA.
INTRODUCTION
Hair loss usually develops gradually and may be patchy or generalized (diffuse) as shown in FIG.1. A person loses about 100 hairs from his head every day. The. Average scalp contains about 150,000 hairs. Each individual hair survives for an average of 3 to 5 years[1]. Generally, the hair falls in its fifth year and is replaced within 6 months by a new one.
Male Pattern hair loss (MPHL), or androgenic alopecia, is the most prevalent types of hair loss. [2],[3] It affects 30% to 50% of men by age 50. Male Pattern hair loss (MPHL) is often regarded as a relatively minor medical condition, but it may result in anxiety and depression in some men because it impacts self-image. (MPHL) caused low self- esteem, depression, and dissatisfaction with body appearance in several study [4,5].
The male pattern baldness involves a line of receding hairline and thinning around the crown with eventual bald spots. In the final stages you can be only one ring hair horseshoe shaped around the sides.
Some women also develop a particular pattern of hair loss due to genetics, age and male hormones (which tend to increase in women after menopause), but this pattern is different from men. Female pattern alopecia involves a thinning throughout the scalp while the frontal hair line generally remains intact hair.
Both men and women the loss of hair thickness and hair count tend to occur as they aged. Alopecia usually is not caused by a disease and is related to aging, heredity and testosterone. Hereditary alopecia or "pattern" affects many more men than women. About 25% of men begin to bald at 30 years old and about two-thirds are either bald or have a balding pattern by age 60.

HAIR LOSS STAGES
Stage 1. No significant hair loss or recession of the hairline.
Stage 2. There is a slight recession of the hairline around the temples. This is also known as an adult or mature hairline.
Stage 3. The first signs of clinically significant balding appear. The hairline becomes deeply recessed at both temples, resembling an M, U, or V shape. The recessed spots are completely bare or sparsely covered in hair.
Stage 4. The hairline recession is more severe than in stage 2, and there is sparse hair or no hair on the vertex. The two areas of hair loss are separated by a band of hair that connects to the hair remaining on the sides of the scalp.
Stage 5. The two areas of hair loss are larger than in stage 4. They are still separated, but the band of hair between them is narrower and sparser.
Stage 6. The balding areas at the temples join with the balding area at the vertex. The band of hair across the top of the head is gone or sparse.
Stage 7. The most severe stage of hair loss, only a band of hair going around the sides of the head remains. This hair is usually not dense and may be fine.
The prevalence of hair loss type III or greater in men 18 to 49 years old has been estimated to be at least 42%. Within the age range of 40 to 49 years, 53% of men have moderate to extensive hair loss. Female pattern hair loss (FPHL) is the most common form of alopecia in women. [6]Affected women may experience psychological distress[4] and impaired social functioning. Twelve percent of women first develop clinically detectable (FPHL) by age 29 years, 25% by age 49 years, 41% by 69 years, and > 50% have some element of (FPHL) by 79 years.
Hair loss in women is polygenic and multifactorial with the additional influence of environmental factors. Several studies focused on the importance of several genes related to alopecia. (FPHL) involves progressive hair follicle miniaturization[7] and subsequently the conversion of terminal follicles into vellus-like follicles [8]. These vellus-like follicles have a shortened hair cycle because of a reduction in the anagen phase, which leads to the production of short and fine hair shafts. Unlike in men, the miniaturization is not uniform and intense in women; therefore, there are no complete areas of baldness except in very rare cases. Moreover, the miniaturization process may be accompanied by a mild-to- moderate lympho-histiocytic inflammatory infiltrate in the peri-infundibular region. Our study assessed the efficacy and safety Of a 5% & 2% BIOHAIR®.[9,10,11,12]
MATERIAL AND METHODS
A phase 2 before-and-after trial was carried out on 50 patients with hair loss for 4-6 months at the Aesthetics Clinic Geneva, Switzerland.
Inclusion criteria were men with (MPHL) between 30 and 70 years old, the female with (FPHL) between 20-50 years with normal health status, and providing written informed consent.
Exclusion criteria were sensitivity to BIOHAIR®, using any topical OTC or prescription medication for hair growth within the past 3 months, using 5α-reductase inhibitors within the past year, using isotretinoin within the past year, radiation to the scalp within the past year, chemotherapy within the past year, using botanicals/nutraceuticals for hair regrowth for the past 3 months, using systemic steroids for more than 14 days within the past 2 months prior to enrollment in the study, uncontrolled hypertension, history of hypotension, any chronic active scalp inflammation or infection, any untreated cancer excluding basal cell carcinoma and squamous cell carcinoma of non-scalp areas, scalp reduction, and use of hair weaves. [13,14]
Trial planning
The subjects were instructed to apply one (2 mL) of BIOHAIR® to the scalp and then massage it into the vertex and frontal balding scalp twice a day and not wash it for at least 6 hours.
Efficacy assessment
The following assessments were made at baseline and at 16 and 24 weeks after treatment:
A) A semi-permanent ink-dot tattoo was placed for precise localization of the target area.
B) A camera was used to take photographs of the target area and the entire scalp in precisely fixed situations.
C) All visible hairs were dot-mapped and counted by a technician trained in the procedure and blinded to the intervention
Subject assessment: Subjects were asked to fill out a questionnaire that rated their overall hair-loss condition in the vertex region compared to baseline. They rated their perception of their hair-loss condition compared to the baseline using a five-point scale, on which -2 = moderately worse, -1 = minimally worse, 0 = no change, +1 = minimally improved, +2 = moderately improved.
Global photographic review (GPR)[15]: GPR was carried out at baseline and at 6 months after treatment. The baseline and post-treatment pictures were shown in a side-by-side presentation and were rated independently by dermatologist using the same five-point scale as above[16].
Safety assessment: The patients were asked and examined for possible side effects, including signs of scalp irritation such as dryness/scaling, folliculitis, and erythema.Data collection and analysis: A specific case report form was prepared and completed for each patient to collect data. Percentage and frequency were used to describe qualitative data, and mean and standard deviation were used for description of quantitative data. The comparison of quantitative data before and after the test was performed by non-parametric equivalent.
Ethics: All patients signed an informed consent form prior to inclusion.
STATISTICAL METHODS
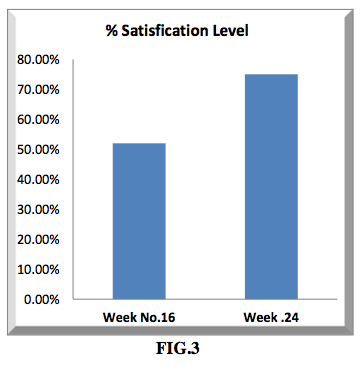
The satisfaction level of the participants was statistically measured in terms of percentage level satisfaction based on the evaluation of hair density and hair growth rate as shown in figure 1 and figure 2 and from that the percentage satisfaction level is drawn as shown in figure 3.
The collection of data were carried out fairly accordingly to the treatment regimen of BIOHAIR® 5% & 2% topical lotion for men and women respectively; the satisfaction level of the volunteers were graphically expressed which shows that at what extent the volunteers were satisfied with the efficacy and safety upon the completion of 24 weeks treatment regimen, table (1).

Results
50 volunteers were enrolled in this study for 16 weeks, and 10 continued up to 24 weeks to assess the treatment response, showed a 78.6% improvement in treatment response. This outcome confirms the result as shown Graphically (FIG.3).
In our study, an expert panel review by global photographic assessment, which is a useful follow-up tool as shown By (FIG.4), (FIG.5), (FIG. 6), (FIG.7), (FIG.8).
Our results showed that the participants were satisfied with the drug efficacy at week 16 (50.2%) SATISFIED and week 24 (78.6%) VERY SATISFIED.The efficacy result was expressed in terms of total number of hair count per square centimeter area and the obtained results checked with a camera after week 16 was (181.87 ±52.42) hair/cm2 and after week 24 (194.58 ± 62.82) hair/cm2 and with an eye count at week 16 (62.57 ± 15.28) hair/cm2 and week 24 (69.91 ± 15.61) hair/cm2 increased significantly compared to the baseline after intervention.
Adverse effects after the use of BIOHAIR® our research results showed that the tolerability profile was high and the low rate of irritant contact dermatitis.[17]

Discussion
Androgenic alopecia is the most common, as it is known as baldness[18]. This generally affects 40% of the population of both men and women in their adult stage. With a higher incidence in men than women. Our results demonstrated that after topical application of Biohair® 5 % & 2% lotion to volunteers with (MPHL) & (FPHL) who were enrolled in this study accordingly to the administration protocol, an overall excellent improvement of hair growth has observed upon the completion of 24 weeks of treatment.
It is well known that the concomitant use of anti-inflammatory steroids is indicated as an aid in hair loss, for treating thinning and hair loss; as well as hereditary baldness both male and female. Stabilizes and stops hair loss. Minoxidil[19] is a potent peripheral vasodilator increase the blood supply to the hair follicle.it is clinically proven to help regrow hair in both men and women increasing hair count and thickness with high tolerable profile.[20]
The combined therapy minoxidil with 0.01% tretinoin and Clobetasol of Biohair® show significant improvement of the Hair count, hair diameter and the hair texture becomes more boosted with high tolerability profile.
Concomitant use with Clobetasol Propionate has substantially increased the effect of Minoxidil, as it acts as an inducer of protein inhibitors phospholipase A2. Clobetasol propionate is a synthetic fluorinated corticosteroid.
Tretinoin used in dermatology, according to their pharmacodynamics, however there is a synergy with Minoxidil. Tretinoin, topically applied inhibits keratinization enhancing terminal hair regrowth.[21]
Tretinoin is minimally absorbed when administered topically. About 80% remains on the skin surface and about 9% and 18% penetrates the stratum corneum, after 10 min and up to 16 hours of contact with the skin, - respectively.
The obtained favorable results shows that Biohair® 5% & 2% is highly effective for all ages (young & adults) patients of MPHL & FPHL. It has found from the clinical study that both Biohair® 5 % & 2% topical lotion are highly effective and safe for the treatment of hair loss Alopecia.[22,23]


DETAILED PHARMACOLOGY PHARMACOKINETICS
Results from a long term dermal toxicity study in rats concluded that there was no apparent relationship between the nadir minoxidl levels and gross cardiac pathology[24].These nadir minoxidil levels in the rat were approximately 40 to 500 times higher than those documented in humans.
Results from a long term dermal toxicity in rabbits concluded that increased heart and liver weights were drug-related; however no Concomitant histopathologic lesions were seen.
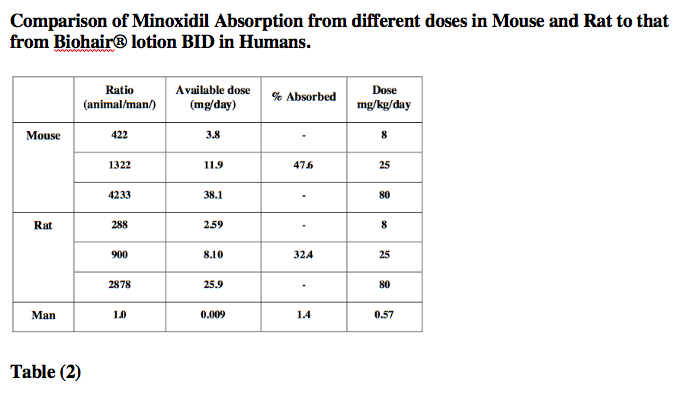
The following table provides a comparison of topically absorbed doses from a 2-year dermal carcinogenicity study in mouse and rat. Compared to results obtained from humans using the recommended twice daily dose of Biohair® lotion. The table demonstrate that on an mg/kg basis, the animals received higher doses than humans. In addition minoxidl in animals (in preclinical toxicity studies) is several hundred times greater than that associated with clinically recommended doses in man.
Following topical application, the urinary metabolite profiles in the rat corresponded closely to those observed after systemic administration, suggesting that metabolism was not altered by entry across the skin.
Characteristics of the topical absorption of 14 C-labelled minoxidil differ appreciably in monkey scalp. During the 10-16 days the monkeys were chaired, approximately 4% of the 1% solution and 1% of the 4% solution appeared in the urine. When returned to metabolism cage, a larger component of total urinary excretion occurred; urinary excretion did not follow first order kinetics. Total absorption in the monkeys was 17.4% of a 1% solution and 5.7% of a 4% solution.
The transdermal metabolism[25] of 14C-minoxidil in fresh human skin in an in-vitro diffusion system was studied. The dermal metabolism of minoxidl in human skin under these in-vitro conditions was minimal at 4.8 to 6% of the applied dose, Table (2).
Extent of Absorption
A four-ways cross-over study in 23 male subjects demonstrated that the contact time of Minoxidil 5% topical lotion affects absorption. Treatment involved dosing of 1mL q12h for 6 days applied to constant
surface area of the scalp. The scalp was washed one, two, four, and 11.5 hours post dose. With increase contact time, absorption increased disproportionately. More than 50% of the minoxidil that is eventually absorbed is absorbed in the first hour post dose, and absorption is nearly complete after 4 hours. [26,27]
PHARMACODYNAMICS
Exploratory in vivo and in vitro studies, designed to determine the mechanism by which Biohair® lotion stimulates hair growth in patient with male pattern baldness have been completed, but have not been successful in definitely demonstrating the mechanism of action of Biohair® % lotion in stimulating hair growth. Studies have shown that there appears to be an immediate vasodilatation of the micro circulation after topical application Biohair® lotion and there is no significant alteration of the effects of androgens on scalp hair. These studies have also shown that cultured epidermal cells appear to be stimulated to divide under the influence of Biohair® % lotion and that in vitro cell cultures of lymphocytes are inhibited in their response to mitogens when Biohair® % lotion monoxide is present in culture.
Results of two studies evaluating minoxidil tablets in doses up to 5 mg twice daily for up to 28 days in normotensive patients show that there were no clinically significant effects on blood pressure measurements or no pulse rate. In addition, there were no clinically significant change in maximum heart response to standard treadmill test, pulse and blood pressure response to a dynamometer grip device, forearm blood flow, plasma rennin level or urine epinephrine and norepinephrine levels. No evidence of fluid retention was seen.
The conclusion was made that low to-moderate, short-term doses of minoxidl tablets, in normotensive volunteers, do not lower blood pressure, and side effects commonly observed during minoxidil tablet therapy for hypertensive patients do not occur in normotensive subjects.
No clinically significant systemic effects were noted in a 16 week, placebo-controlled, randomized study of 98 treated hypertensive pateints9involving B-blockers, diuretics) who were also treated with Biohair® 5 % topical lotion.
Untreated hypertensive patients were evaluated in an IV minoxidil study. The subject achieved pharmacokinetics steady-state within 6 hours after the start of infusion. The heart rate and diastolic blood pressure effects observed indicated that IV infusion of 1.37 mg and 3.43 mg minoxidil did not result in clinically significant cardiovascular effects. The 6.686 mg dose, which resulted in a mean serum minoxidil concentration of 21.7ng/mL, was the lowest clearly distinguishable from placebo, based on heart rate data.
Conclusion
Hair loss is one of the most prevalent dermatological illnesses that cause patients to seek treatment. There are limited options for treating it effectively. This is why hair loss remains an important area for further research to obtain more information regarding its pathogenesis and newer therapeutic options that are now being developed. The study indicates that BIOHAIR® is highly effective treatment for Alopecia in men and Women it can increase the growth of new hairs count and density. The study indicates that BIOHAIR® is Safe in Men and women.
The daily administration of a proprietary BIOHAIR® significantly increased hair growth, it takes to 24 weeks to produce visible result as it has shown in this clinical study report.
Together, this study will help provide a more detailed understanding of the mechanism of this new drug to promote hair growth and add to the existing body of clinical evidence.
References:
1. Stene, J.J., [Hair physiology]. Rev Med Brux, 2004. 25(4): p. A263-5.
2. Wessagowit, V., et al., Treatment of male androgenetic alopecia with topical products
containing Serenoa repens extract. Australas J Dermatol, 2016. 57(3): p. e76-82.
3. Lee, H.H., et al., Epidemiology of alopecia areata, ophiasis, totalis and universalis: A systematic review and meta-analysis. J Am Acad Dermatol, 2019.
4. Cash, T.F., The psychological effects of androgenetic alopecia in men. J Am Acad Dermatol, 1992. 26(6): p. 926-31.
5. Cash, T.F., V.H. Price, and R.C. Savin, Psychological effects of androgenetic alopecia on women: comparisons with balding men and with female control subjects. J Am Acad Dermatol, 1993. 29(4): p. 568-75.
6. Conic, R.R., S. Khetarpal, and W. Bergfeld, Treatment of female pattern hair loss with combination therapy. Semin Cutan Med Surg, 2018. 37(4): p. 247-253.
7. Whiting, D.A., Possible mechanisms of miniaturization during androgenetic alopecia or pattern hair loss. J Am Acad Dermatol, 2001. 45(3 Suppl): p. S81-6.
8. Herskovitz, I., I.C. de Sousa, and A. Tosti, Vellus hairs in the frontal scalp in early female pattern hair loss. Int J Trichology, 2013. 5(3): p. 118-20.
9. Olsen, E.A., et al., A randomized clinical trial of 5% topical minoxidil versus 2% topical minoxidil and placebo in the treatment of androgenetic alopecia in men. J Am Acad Dermatol, 2002. 47(3): p. 377-85.
10. Sharma, A., et al., Tretinoin enhances minoxidil response in androgenetic alopecia patients by upregulating follicular sulfotransferase enzymes. Dermatol Ther, 2019. 32(3): p. e12915.
11. Tosti, A., et al., Clobetasol propionate 0.05% under occlusion in the treatment of alopecia totalis/universalis. J Am Acad Dermatol, 2003. 49(1): p. 96-8.
12. Sung, C.T., et al., The Efficacy of Topical Minoxidil for Non-Scarring Alopecia: A Systematic Review. J Drugs Dermatol, 2019. 18(2): p. 155-160.
13. Sasaki, G.H., Review of Human Hair Follicle Biology: Dynamics of Niches and Stem Cell Regulation for Possible Therapeutic Hair Stimulation for Plastic Surgeons. Aesthetic Plast Surg, 2019. 43(1): p. 253-266.
14. Azar, R.P., et al., Asymmetry of the Receding Hairline in Men With Early Androgenetic Alopecia. J Cutan Med Surg, 2016. 20(6): p. 546-549.
15. Dhurat, R. and P. Saraogi, Hair evaluation methods: merits and demerits. Int J Trichology, 2009. 1(2): p. 108-19.
16. Neste Dominique, V., Photographic Techniques and Quantification of Hair Growth. 2017. p. 767-786.
17. Keaney, T.C., et al., Efficacy and Safety of Minoxidil 5% Foam in Combination With a Botanical Hair Solution in Men With Androgenic Alopecia. J Drugs Dermatol, 2016. 15(4): p. 406-12.
18. Hoffmann, R., Male androgenetic alopecia. Clin Exp Dermatol, 2002. 27(5): p. 373-82.
19. Topical minoxidil approved by FDA. Clin Pharm, 1988. 7(12): p. 858, 862.
20. Ghonemy, S., H. Bessar, and A. Alarawi, Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic evaluation. J Dermatolog Treat, 2019: p. 1-24.
21. Bazzano, G.S., N. Terezakis, and W. Galen, Topical tretinoin for hair growth promotion. J Am Acad Dermatol, 1986. 15(4 Pt 2): p. 880-3, 890-3.
22. Shin, H.S., et al., Efficacy of 5% minoxidil versus combined 5% minoxidil and 0.01% tretinoin for male pattern hair loss: a randomized, double-blind, comparative clinical trial. Am J Clin Dermatol, 2007. 8(5): p. 285-90.
23. Gajjar, P.C., et al., Comparative Study between Mesotherapy and Topical 5% Minoxidil by Dermoscopic Evaluation for Androgenic Alopecia in Male: A Randomized Controlled Trial. Int J Trichology, 2019. 11(2): p. 58-67.
24. Gottlieb, T.B., R.C. Thomas, and C.A. Chidsey, Pharmacokinetic studies of minoxidil. Clin Pharmacol Ther, 1972. 13(3): p. 436-41.
25. Goren, A., et al., The effect of topical minoxidil treatment on follicular sulfotransferase enzymatic activity. J Biol Regul Homeost Agents, 2018. 32(4): p. 937-940.
26. Ferry, J.J., J.H. Shepard, and G.J. Szpunar, Relationship between contact time of applied dose and percutaneous absorption of minoxidil from a topical solution. J Pharm Sci, 1990. 79(6): p. 483-6.
27. Latriano, L., et al., The percutaneous absorption of topically applied tretinoin and its effect on endogenous concentrations of tretinoin and its metabolites after single doses or long-term use. J Am Acad Dermatol, 1997. 36(3 Pt 2): p. S37-46.
Помеченный: Восстановление волос
Rate this article
Share this article on


